Product name: Eurycomanone
Synonym name:
Catalogue No.: BP1784
Cas No.: 84633-29-4
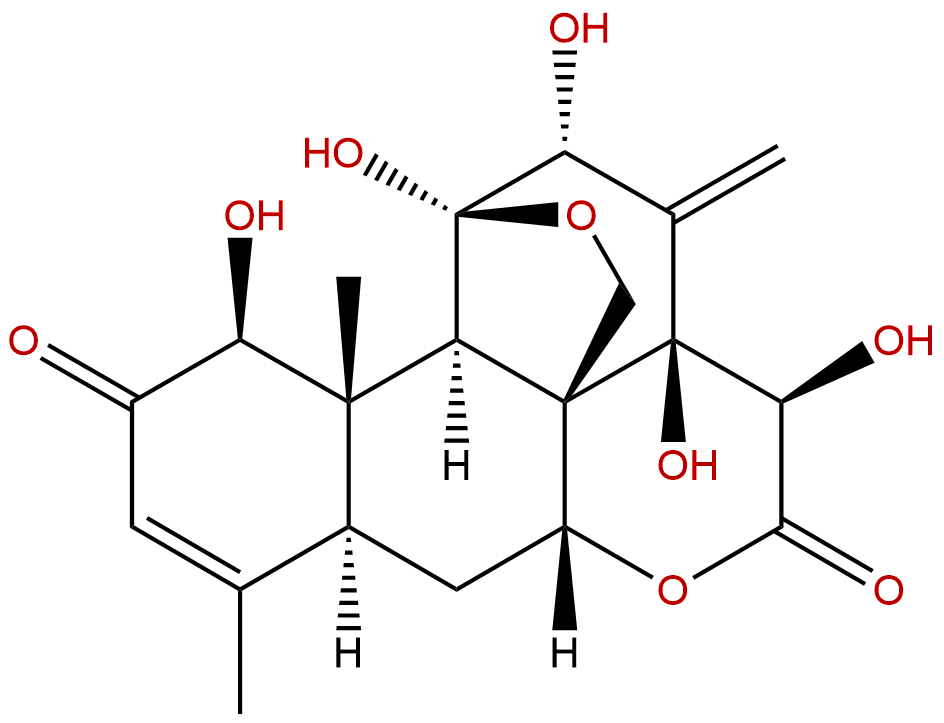
Formula: C20H24O9
Mol Weight: 408.403
Botanical Source: Eurycoma longifolia
Physical Description: Cryst.
Type of Compound: Diterpenoids
Purity: 95%~99%
Analysis Method: HPLC-DAD or/and HPLC-ELSD
Identification Method: Mass, NMR
Packing: Brown vial or HDPE plastic bottle
Storage: Store in a well closed container, protected from air and light. Put into refrigerate or freeze for long term storage.
Whenever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20℃. Generally, these will be useable for up to two weeks.
The product could be supplied from milligrams to grams, up to kilograms
Inquire for bulk scale.
Descriptions:
Eurycomanone has cytotoxic on HepG2 cells and Human Cervical Carcinoma Cells by inducing apoptosis through the up-regulation of p53 and Bax, and down-regulation of Bcl-2.[1,2]
Eurycomanone exerts antiproliferative activity via apoptosis in Hela cells MCF-7cells .[3,4] and
Eurycomanone at viable therapeutic concentrations of 5–20μg/ml exhibits significant anti-proliferative and anti-clonogenic cell growth effects on A549 lung cancer cells, the treatment also resulted in suppression of the lung cancer cell tumor markers and several known cancer cell growth-associated genes.[5]
Eurycomanone enhances testosterone steroidogenesis at the Leydig cells by inhibiting aromatase conversion of testosterone to oestrogen, and at a high concentration may also involve phosphodiesterase inhibition, it may be worthy for further development as a phytomedicine to treat testosterone-deficient idiopathic male infertility and sterility.[6]
Eurycomanone and eurycomanol as regulators of signaling pathways involved in proliferation, cell death and inflammation.[7]
Eurycomanone possesses growth inhibition of P. berghei by combination of eurycomanone-artesunate with doses 30 mg/kgBW-artesunate 4mg/kgBW, suggests that this combination can be used as potential antimalarial drug.[8]
References:
[1] Zakaria Y, Rahmat A, Pihie A H L, et al. Cancer Cell Int, 2009, 9(1):1-21.
[2] Mahfudh N, Pihie A H L. J Cancer Mol, 2008, 4(4):109-15.
[3] Nurkhasanah, Azimahtol Hawariah Lope Pihie,Jalifah Latip. Majalah Farmasi Indonesia, 2009, 20(4): 190-7.
[4] Pihie A H L. Anticancer Res, 2004, 24(5D):3426-7.
[5] Wong P F, Cheong W F, Shu M H, et al.Phytomedicine, 2012, 19(2):138-44.
[6] Low B S, Choi S B, Wahab H A, et al. J Ethnopharmacol, 2013, 149(1):201-7.
[7] Hajjouli S, Chateauvieux S, Teiten M H, et al. Molecules, 2014, 19(9):14649-66.
[8] Yusuf H, Satria D. J Chem Pharm Res, 2016,(82):18-22.
[9] Khari N, Aisha A, Ismail Z. Trop J Pharm Res, 2014, 13(5):801-7.
Parameter information
Product name:Eurycomanone
CAS No.:84633-29-4
MF:C20H24O9
MW:408.403
EINECS:
Chinese Synonym:
Name:Eurycomanone
Synonym:
- Purity/Standard:98%
-
No.:BP1784-100mg
Packing:100mg
Brand:Phytounique
Stock:In stock$ 420.00
$ 420.00 Put into shopping cart - Purity/Standard:98%
-
No.:BP1784-20mg
Packing:20mg
Brand:Phytounique
Stock:In stock$ 150.00
$ 150.00 Put into shopping cart - Purity/Standard:98%
-
No.:BP1784-10mg
Packing:10mg
Brand:Phytounique
Stock:In stock$ 96.00
$ 96.00 Put into shopping cart
- relateproduct